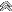How To: Correct Antifreeze Analysis/Mixture
In a perfect world, if I have a radiator that is filled with a 50/50 mix of antifreeze and water and I drain approximately 3 gallons after pulling the lower radiator hose, then what is the percentage left in the cooling system and how many gallons of antifreeze will I need? A logical answer would be 1.5 gallons of antifreeze and 1.5 gallons of distilled water right?
The answer is YES (in a perfect world). If you know you already have a good 50/50 mix and you tested it, then you will likely be 'on the money' with your coolant mix. But what if you didn't start with a 50/50? or what if you wanted your truck to have a 60/40 or 70/30 mixture? Or going further, what if you weren't sure how much antifreeze you had drained/lost during a repair? I created this chart so that I could change my mixture on the fly, simply by using a cheap antifreeze tester. According to Zerex Technical Bulletin (the ELC that I used for my 6.0) maximum freezepoint is -90 degrees Fahrenheit and boiling point is 277 degrees Fahrenheit. This chart reflects the average for most Ethylene Glycol coolants. You will notice that my chart doesn't go down to a -90 deg F temp. So this isn't precise temp chart, but it doesn't matter because I haven't found a coolant tester that can even measure mixtures below -45 degrees anyway.

Enough of that, let's get to the testing.
1. Start by draining enough anti-freeze through the radiator draincock to suck up into the tester. Approximately 1/2 cup should be sufficient. If you have enough fluid in the degas, you can put the tester inside it and suck it up. I found that if I run my anti-freeze at 1/2" below the min line on the degas, it is not very easy to get the tester far enough in to suck up the fluid, so I resorted to the drain method.
I purchased a cheap antifreeze tester. There is a specific way to test using this particular tester. To get an accurate test result, you must do the following:
1. The arrow at the top must be centered in the crosshairs to get an accurate reading.

2. The max fluid level line should not be exceeded.

My first result was a reading of +5 degrees Fahrenheit.

Looking at the chart, I see that this means I have an approximate mix of 29% Antifreeze and 71% distilled water. (THIS IS NOT GOOD! This means that my mixture will freeze when the weather drops to +5 degrees F.
So what do you do? How much do you drain and fill? Anyone like to guess? I decided to test the mixture by draining exactly 1 gallon of fluid from my radiator and adding 1 Gallon of full strength (undiluted) Zerex ELC to my cooling system. The result was a reading of (minus) -10 degrees Fahrenheit. Looking at the chart again, I see that this means I have a mixture of 39% antifreeze and 61% distilled water.

This is also not a good mixture. (negative) -10 degrees may work for me in the South, but I wouldn't want to risk it, and my system is also missing out on anti-rust agents, lubricants for the water pump and an anti-foaming agent that is in the coolant.
Doing the math (this is based on the same equation I used in the chart), how do we only go from 29% antifreeze to 39% antifreeze when draining a gallon of pre-mix and adding a gallon of full strength antifreeze? In other words, the question is:
An F250 radiator holds 28 quarts of fluid (7 gallons). There is currently a mixture in the radiator that is 29% antifreeze. How much of this mixture should be drained and replaced by pure antifreeze so that the resulting mixture is 39% antifreeze?
Answer:
Current mix =29%
Capacity is 28 quarts
Target mix =39%
<table border="0" cellpadding="0" cellspacing="0" width="314"><colgroup><col style="mso-width-source:userset;mso-width-alt:11483;width:236pt" width="314"> </colgroup><tbody><tr style="height:15.0pt" height="20"> <td style="height:15.0pt;width:236pt" height="20" width="314">(29%)(28-x)+(100%)(x)=(39%)(28)</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">(.29)(28-x)+x=.39(28)</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">8.12 - .29x + x = 10.92</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">(-.29x) + 1x = 10.92 - 8.12</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">.71x = 2.8</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">x = 2.8/.71</td> </tr> <tr style="height:15.0pt" height="20"> <td style="height:15.0pt" height="20">x = 3.9436 quarts</td> </tr> </tbody></table>
So you can see from our math that we needed to add 3.9436 qts in order to take the mixture from 29% to 39%, so this proves that the math is very close to real life scenario. How do we know? Well, I added a gallon (4 quarts), and was able to increase my freeze point from a +5 F on my coolant tester, to a -10 F reading. 4 quarts is very close to 3.9436 quarts from our equation right?
So I'm sure while you are looking at this, you're thinking "What did you prove by going from 29% to 39%? The answer is that I needed a sanity check. I'm not a chemist so I needed to prove that this chart would be correct for all scenarios other than just 'theory' on a piece of paper. I needed to prove that it could be applied to our cooling systems.
So let's take a close look at our chart... How do we read it?:

Using our first scenario above. How do we go from a +5 deg F freezepoint reading, directly to a proper 50/50 mixture?
1. First we take the reading from our coolant tester.
2. Next, we scroll over to the right until we hit the column for our target mixture. In this case, we want a 50/50 mix.
3. The result is that we need to drain 8.28 quarts and refill with 8.28 quarts of pure, undiluted antifreeze.
Chart reading is below:

Let's go through another scenario to make sure that we are reading the chart correctly.
Question: If my current freezpoint reading is negative -10 deg. Fahrenheit, how much pure/undiluted antifreeze will I need to drain and add if I want a 59% Antifreeze and 41% distilled water mixture?
Answer: 11.48 quarts or approximately 3 gallons needs to be drained from my cooling system and 3 gallons added. This gives me a freezepoint protection of negative -60 deg F and a boiling point of 270 deg Fahrenheit.
So before you decide "Hey, I'll just run 70% antifreeze and 30% distilled water for the max freezepoint and boiling point protection." Just remember, antifreeze/coolant is a chemical modifier and it actually has poor heat transfer properties. It functions to change the boiling and freezing points of water. It also includes anti-rust agents, lubricants for the water pump and an anti-foaming agent. It is water that does the work of cooling and the proper mix of antifreeze/coolant is there to help the water work better.
So at this point, you're thinking "Hey, why not just fill the entire cooling system with Antifreeze?" Well, believe it or not, antifreeze/coolant has a higher freeze point than water when it is full strength. Anywhere from 0 to +10 from what I have seen listed for Ethylene Glycol.
Hope this helps you guys. PS. I can change the Capacity in my excel sheet and it will work for any cooling system.
1. What is the recommended mixture for 6.0 Powerstroke?
Unless you are in an extremely cold climate, 50/50 will work practically anywhere. -34 deg F. freezepoint and 265 deg F. boiling point with a 16psi cap.
2. How important is the 16 psi degas bottle cap?
First, the radiator cap is an important part of the cooling system. It is designed to hold a certain amount of pressure inside the system, and to release the pressure when it exceeds a pre-determined level. Why do you want pressure in the cooling system? Actually, you don’t get a choice. As the coolant heats up, it expands. Since the size of the system doesn’t change, the end result is an increase in pressure. However, the positive impact is that the boiling point of the coolant increases as the pressure increases. Straight water at atmospheric pressure boils at 212 degrees Fahrenheit. A mix of 50/50 coolant and water boils at around 220 degrees. That same mix at 16 pounds per square inch (psi) of pressure boils at approximately 250 degrees. And the boiling point increases 2-3 degrees for each additional psi of pressure in the system.
3. I tried using the chart, but it didn't work. What did I do wrong?
It is important to remember that if the chart shows 8.28 quarts for your target, this means that you MUST drain ALL 8.28 quarts at one time. You can't drain one gallon, then fill your system with a gallon of antifreeze and then drain the system again, and fill with another 1 gallon and 9 ounces. Think about it, if you take a mixture that is 29% antifreeze and you drain it, you are actually losing a percentage of pure antifreeze. The chart will ONLY work if you follow the exact drain amount and re-fill with full strength/undliluted antifreeze.
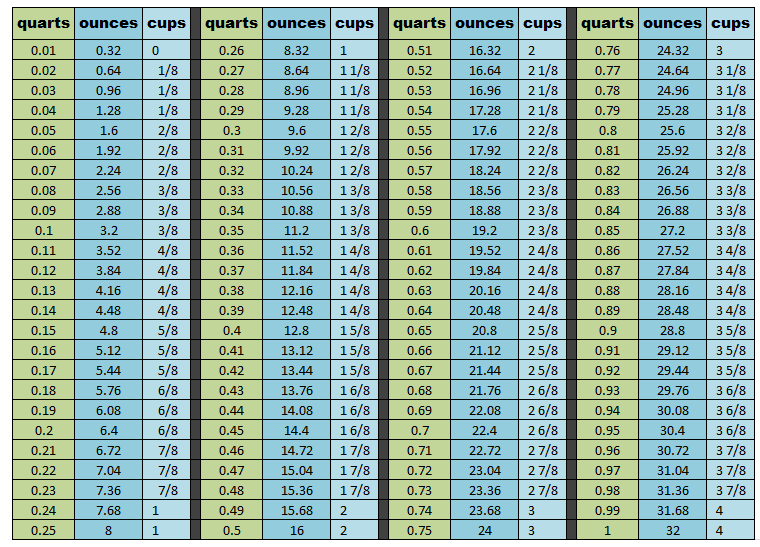
4. You mentioned 8.28 quarts in your answer above. How much is .28 quarts? How can I measure that?
I used this conversion chart. Keep in mind that the cups are rounded up to the nearest 1/8 of a cup. According to the chart below .28 quarts = 8.96 ounces = 1 and 1/8 cup.
* NO, you DO NOT have to be that precise, but I will leave it up to the owner whether he/she decides to round down or up when filling their cooling system.
 I thought about using less words but felt that it's better to explain it all up front rather than have question after question.
I thought about using less words but felt that it's better to explain it all up front rather than have question after question.