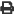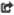Oh no my aluminum is rusting badly
#2
#5
Bare Aluminum rapidly forms a passivating layer of Aluminum Oxide. Unlike Ferric Oxide (our ol' buddy, rust), Aluminum Oxide protects the underlying Aluminum. Unless it's oxide is removed and prevented form re-forming, then it goes quickly!
And in design, there is the whole galvanic action issue. There were full-size Fords and Mercs some years that had aluminum rear bumpers, the big ones. They had two isolation pads that fit in between the mounting areas of the bumper, and the two steel frame-ends which were the bumper mounts. They were, or were like, lead sheets. They stuck out way beyond the joint on all 4 sides.
And in design, there is the whole galvanic action issue. There were full-size Fords and Mercs some years that had aluminum rear bumpers, the big ones. They had two isolation pads that fit in between the mounting areas of the bumper, and the two steel frame-ends which were the bumper mounts. They were, or were like, lead sheets. They stuck out way beyond the joint on all 4 sides.
#7
Trending Topics
#8
#9
#11
sodium chloride (NaCl), aka salt, dissolves and lowers the freezing point of water, higher concentration = lower freezing point.
calcium chloride(CaCl), aka ice melt, reacts with water and generates a small amount of heat and melts ice and snow.
with a mixture, the CaCl will melt snow and ice,the NaCl will mix with the water and lower the freezing point. I think MgCl can be used the same as CaCl but not sure.
All are corrosive to metals.
calcium chloride(CaCl), aka ice melt, reacts with water and generates a small amount of heat and melts ice and snow.
with a mixture, the CaCl will melt snow and ice,the NaCl will mix with the water and lower the freezing point. I think MgCl can be used the same as CaCl but not sure.
All are corrosive to metals.
#13
Bare Aluminum rapidly forms a passivating layer of Aluminum Oxide. Unlike Ferric Oxide (our ol' buddy, rust), Aluminum Oxide protects the underlying Aluminum. Unless it's oxide is removed and prevented form re-forming, then it goes quickly!
And in design, there is the whole galvanic action issue. There were full-size Fords and Mercs some years that had aluminum rear bumpers, the big ones. They had two isolation pads that fit in between the mounting areas of the bumper, and the two steel frame-ends which were the bumper mounts. They were, or were like, lead sheets. They stuck out way beyond the joint on all 4 sides.
And in design, there is the whole galvanic action issue. There were full-size Fords and Mercs some years that had aluminum rear bumpers, the big ones. They had two isolation pads that fit in between the mounting areas of the bumper, and the two steel frame-ends which were the bumper mounts. They were, or were like, lead sheets. They stuck out way beyond the joint on all 4 sides.
Go find a three or four year old Explorer or Mustang and check out the leading edge of their aluminum hood. Virtually every one I've seen has the paint blistering and peeling in several spots. Apparently the problem is that the hood skin is aluminum but the stiffening ribs are steel. One rock chip on the leading edge and the cancer starts.